Meningitis: Symptoms, Causes, Vaccines and the UK Protection Gap
Meningitis is a serious infection that can progress rapidly and requires urgent medical treatment. This in-depth guide explains the causes, symptoms, vaccines, and the current protection gap in the UK.

Meningitis kills quickly. It can leave survivors with profound disability. And it remains widely misunderstood, even by people who think they know the warning signs. The glass test, the rash, the stiff neck: these are the things most people associate with meningitis, but they are often late signs, and in many cases they never appear at all.
This post does not set out to be a symptom guide. It sets out to explain what meningitis actually is, why it is more varied and more complicated than most awareness content acknowledges, and why a cohort of children and young people in the UK are currently moving through their adolescence with less protection than they need.
That last point is not a criticism of policy. It is a description of where things stand, and understanding it matters for anyone who wants to make genuinely informed decisions about the people in their care.
If you are looking for a quick practical guide to recognising meningitis in teenagers, see our guide for parents.
What Is Meningitis?
Meningitis is inflammation of the meninges: the three protective membranes, called the dura mater, arachnoid mater, and pia mater, that surround the brain and spinal cord. These membranes serve as a physical barrier between the central nervous system and everything outside it. When they become inflamed, the consequences can be severe.
The inflammation is usually caused by infection, most commonly viral or bacterial. Rarer causes include fungi, parasites, certain medications, and the brain's own immune response in autoimmune conditions. The cause matters enormously, because it determines how serious the illness is, how it is treated, and what the likely outcome will be.
Meningitis is often discussed alongside septicaemia (blood poisoning). These are related but distinct conditions. Meningitis describes infection of the meninges. Septicaemia describes infection that has spread into the bloodstream. In meningococcal disease, caused by the bacterium Neisseria meningitidis, these two conditions frequently occur together, but not always. A person can have meningococcal meningitis without septicaemia, or septicaemia without significant meningitis. Understanding this distinction matters because some of the most recognisable symptoms of meningococcal disease, including the non-fading rash, are signs of septicaemia rather than meningitis itself.
Types of Meningitis
The single most important distinction is between bacterial and viral meningitis. They share symptoms, particularly in their early stages, but they differ substantially in severity, treatment, and prognosis.
Viral Meningitis
Viral meningitis is the more common form. The majority of cases resolve without specific treatment, though recovery can take several weeks and fatigue and headaches may persist for months.
The most common cause is enteroviruses, a large family of viruses that includes coxsackieviruses and echoviruses. Enteroviruses circulate widely, particularly in late summer and autumn. Other viral causes include herpes simplex virus (HSV), mumps virus, and HIV in early infection. A national surveillance study of viral meningitis in England between 2013 and 2023 identified enteroviruses as the dominant cause throughout the period.[1]
Viral meningitis does not respond to antibiotics. Treatment is supportive: rest, fluids, and pain relief. However, herpes simplex meningitis is treated with the antiviral aciclovir, making rapid identification important. No clinician or patient should attempt to diagnose the cause of meningitis without investigation. The early symptoms of viral and bacterial meningitis are frequently indistinguishable, and bacterial meningitis can present initially as a mild illness.
Bacterial Meningitis
Bacterial meningitis is less common but far more dangerous. It progresses faster, kills more often, and is more likely to leave survivors with permanent disability.
The most significant bacterial cause in the UK is Neisseria meningitidis, commonly called the meningococcus. Other important causes include Streptococcus pneumoniae (the pneumococcus), which tends to cause more severe disease in adults and older people; Haemophilus influenzae type b (Hib), which was the most common cause in young children before vaccination; Listeria monocytogenes, which disproportionately affects pregnant women, newborns, and the elderly; and Group B Streptococcus, a significant cause in newborns.
Bacterial meningitis requires emergency treatment with intravenous antibiotics. If meningococcal disease is suspected in the community, benzylpenicillin may be given before hospital transfer if this can be done immediately and without delaying urgent transport.
Fungal Meningitis
Fungal meningitis is rare in people with healthy immune systems. The most significant cause is Cryptococcus neoformans, which is a major cause of mortality in people with HIV/AIDS or other severe immunosuppression, particularly in sub-Saharan Africa. Tuberculous meningitis, caused by Mycobacterium tuberculosis, has a more gradual onset than bacterial meningitis and presents its own diagnostic challenges.
Amoebic Meningitis: Primary Amoebic Meningoencephalitis
Most discussions of meningitis focus on viral and bacterial causes, and rightly so. But meningitis can be caused by other organisms, and among these, the free-living amoeba Naegleria fowleri is in a category of its own. Not because of its frequency, but because of what it does and how reliably it does it.
What Is Naegleria fowleri?
Naegleria fowleri is a thermophilic amoeba found in warm freshwater environments: lakes, rivers, hot springs, geothermally heated natural pools, and poorly maintained swimming pools with inadequate chlorination. It does not require a human host. When it enters a human body, it is not behaving as a parasite in any evolutionary sense. It is going somewhere it does not belong, and the consequences are catastrophic.[11]
The infection it causes is called primary amoebic meningoencephalitis, or PAM. It enters the body through the nose, most commonly during water activities where water is forcefully pushed into the nasal passages: diving, jumping into water, or vigorous play in warm freshwater. Swallowing contaminated water does not cause infection.[11]
Once in the nasal passages, the amoeba attaches to the olfactory mucosa, crosses the cribriform plate, the bony structure at the base of the skull through which the olfactory nerves pass, and invades the brain tissue. It destroys brain tissue through direct cell damage and the secretion of cytolytic enzymes. The disease is fatal in more than 98% of cases, with a median survival of around five days from symptom onset.[11]
Symptoms and the Diagnostic Problem
Early symptoms of PAM are largely indistinguishable from bacterial meningitis: sudden severe headache, high fever, nausea, vomiting, and stiff neck. Neurological symptoms follow rapidly, including confusion, hallucinations, seizures, and loss of balance. PAM is so rare that it is usually not on the list of differential diagnoses when a patient presents. CSF examination resembles bacterial meningitis. The amoeba can be identified under microscope from CSF samples, but this requires a specific protocol that most routine laboratories do not use. New Zealand public health guidance is explicit: diagnosis is often made at autopsy.[12]
There is no established standard treatment. Combinations of antifungals, particularly amphotericin B, with antiparasitic agents have been used. The handful of documented survivors globally received aggressive multi-drug therapy initiated very early. The obstacle is not treatment but diagnosis.[11]
The New Zealand Connection
New Zealand has a historically significant relationship with both PAM and meningococcal B disease, and the two are connected in ways that are not widely known.
PAM was first recognised as a clinical entity in New Zealand in 1968, following an outbreak of fatal cases among people swimming in geothermally heated freshwater pools in the central North Island. Eight fatal cases occurred in New Zealand between 1968 and 1984, with a further confirmed death in 2000.[12] The thermal pools of the Rotorua region and the central volcanic plateau, where water temperatures can reach blood temperature or above, provided ideal conditions. The New Zealand cases were among the earliest and most thoroughly documented in the world.
The MenB connection is equally direct. One of the key components of the Bexsero vaccine, its outer membrane vesicle (OMV) component, was derived from a New Zealand MenB outbreak strain. New Zealand experienced a prolonged and severe epidemic of meningococcal group B disease in the late 1980s and 1990s, which led to the development of a New Zealand-specific MenB vaccine deployed between 2004 and 2008. The OMV from that outbreak strain was subsequently incorporated into the development of Bexsero, giving it broader strain coverage.[3] The UK's MenB vaccine owes part of its structure to the lessons learned from a New Zealand public health emergency.
Risk in Context
Fewer than 400 cases of PAM have been documented worldwide. The United States accounts for over 40% of reported cases. Cases in the UK have been exceptionally rare, typically associated with travel to tropical freshwater environments. The actual global figure is likely higher than reported, due to underdiagnosis.[11]
The appropriate response to PAM is not fear of swimming. If you are swimming in warm freshwater, particularly in geothermally active regions or tropical countries, the risk is small but not zero. Noseclips reduce it substantially. The risk from properly maintained, adequately chlorinated pools is negligible. The reason PAM deserves serious treatment in any discussion of meningitis is that it represents the extreme end of a diagnostic problem that runs through all forms of the disease: symptoms that resemble common conditions, a disease that progresses faster than the diagnostic process, and an outcome that is almost invariably fatal without prompt, correct treatment.
The Meningococcus in Detail: Serogroups and Why They Matter
Of all bacterial causes of meningitis in the UK, Neisseria meningitidis warrants particular attention. It is the leading cause of bacterial meningitis in children and young people, it can progress from early symptoms to death within 24 hours, and it is the bacterium at the centre of the UK's ongoing vaccination debates.
The meningococcus is divided into serogroups based on the chemical structure of its polysaccharide capsule. There are at least 12 serogroups, of which five, referred to as A, B, C, W, and Y, cause the overwhelming majority of human disease worldwide.
Serogroup B
MenB is the dominant serogroup in the UK. According to the UKHSA's annual surveillance report for the 2024/25 epidemiological year, it accounted for 82.6% of all confirmed invasive meningococcal disease cases in England.[2] It has the highest disease burden among infants under one year of age, and a second peak in teenagers and young adults aged 15 to 24.
Developing a vaccine against MenB took decades longer than vaccines against other serogroups. The reason is structural: the MenB polysaccharide capsule is chemically similar to molecules found on the surface of human neural cells, making it poorly immunogenic. A vaccine targeting the capsule directly risked triggering an autoimmune response. The breakthrough came through reverse vaccinology: researchers analysed the MenB genome to identify surface proteins that could generate a protective immune response without targeting the capsule itself.[3]
The resulting vaccine, Bexsero, was licensed in Europe in 2013 and added to the NHS routine infant schedule in September 2015, making the UK the first country in the world to introduce it nationally. Surveillance data indicate it covers the substantial majority of MenB strains currently causing invasive disease in England, though not all strains.[2][3]
Serogroup C
MenC was a major cause of disease in the UK in the late 1990s. The introduction of the MenC conjugate vaccine in 1999 reduced group C cases by approximately 96%,[4] one of the most effective vaccination programmes in UK public health history. MenC now accounts for a small minority of cases.
Serogroups A, W, and Y
These three serogroups are covered by the MenACWY vaccine offered to teenagers. Group W disease rose sharply from 2009 onwards, driven by a particularly virulent clone, the MenW ST-11 clonal complex, which caused increasing numbers of deaths. The teenage MenACWY programme introduced in response substantially reduced group W disease: from over 200 cases per year at its peak to 43 in 2024/25.[2] Group A is rare in the UK but causes large epidemics in sub-Saharan Africa's meningitis belt.
Carriage: Why Teenagers Are at Higher Risk
The meningococcus often lives harmlessly in the back of the throat. In the general population, approximately one in ten people carry the bacterium at any given time without symptoms. In teenagers and young adults, that figure rises to around one in four.[5] Carriage is how the bacterium spreads: through close contact, kissing, and shared drinking vessels.
For the vast majority of carriers, nothing happens. In a small number of cases, for reasons not fully understood, the bacterium crosses the mucosal barrier and enters the bloodstream, becoming invasive. Understanding carriage explains why university campuses and other adolescent social environments can allow meningococcal disease to spread quickly.
One critical distinction: the MenB vaccine does not reduce carriage of the bacterium, and therefore provides no herd protection to unvaccinated people around the vaccinated individual. Protection is confined to the person vaccinated. This is unlike the MenACWY vaccine, which does reduce carriage of the groups it covers and therefore offers a degree of indirect protection to the wider community.[2] This difference in mechanism is one reason the public health calculus around extending MenB vaccination is more complicated than it might initially appear.
The Vaccination Gap: Who Is Unprotected and Why
This is the structural problem that defines the current landscape of meningococcal risk in the UK, and it is insufficiently understood outside specialist circles.
The 2015 Cut-off
The MenB infant programme began on 1 September 2015. Only children born on or after 1 May 2015 were eligible for the routine programme.[6] Any child born before that date was not eligible for NHS MenB vaccination unless they fell into a specific clinical risk group.
In practical terms, everyone currently aged ten or over has never been routinely offered MenB vaccination on the NHS. They could have received it privately, but the majority have not. As of 2026, that group includes virtually all secondary school pupils, all university students, and all adults.
This was not an oversight. The JCVI, the independent body that advises UK governments on vaccination, determined that extending MenB vaccination to older children and teenagers did not meet the cost-effectiveness threshold at the time the infant programme was introduced. That is a legitimate policy judgement. Its consequence, a decade on, is an entire generation of adolescents moving through the highest-risk period of meningococcal disease without MenB protection.
The Adolescent Risk Peak
Meningococcal disease has a bimodal age distribution. Risk is highest in infants under one year of age, drops through childhood, then rises again in teenagers and young adults, peaking around ages 15 to 24.[5] This second peak is driven by increased social mixing, higher carriage rates, and possibly changes in the mucosal immune environment during adolescence.
The infants who were the first cohort to receive MenB vaccination in 2015 will turn eleven in 2026. By 2030, they will be entering the adolescent risk peak. Whether the protection they received as babies will remain meaningful at that point is uncertain. Current evidence suggests that vaccine-induced immunity from Bexsero wanes, and that protection in teenagers from infant vaccination alone is unlikely to remain robust.[7] Meningitis Now's 'No Plan B for MenB' campaign has specifically called for an adolescent booster programme to be in place by 2030 to address this.[7]
The MenACWY Picture
Teenagers are offered MenACWY vaccination in Year 9, typically at age 13 to 14. Coverage reached 72.1% in the 2023/24 academic year, better than the post-pandemic low but still well below the pre-pandemic level of 88%.[2] Catch-up vaccination is available to anyone under 25 who missed it. But MenACWY does not cover group B, which now accounts for more than four in five invasive meningococcal cases in England. Teenagers who have received MenACWY are not protected against the most common strain.
Born before May 2015: not routinely offered MenB vaccine on the NHS. This covers all current secondary school and university students.
MenB is now the dominant strain: 82.6% of invasive meningococcal cases in England in 2024/25, according to UKHSA.
MenACWY is offered at school but does not protect against MenB. Coverage is 72%, leaving roughly one in four teenagers without any meningococcal ACWY protection either.
Private MenB Vaccination: A Practical Guide
For anyone born before May 2015, NHS MenB vaccination is not available unless they fall into a clinical risk group. Private vaccination is the only route.
Both licensed MenB vaccines are available privately in the UK: Bexsero (manufactured by GSK) and Trumenba (manufactured by Pfizer). For anyone over two years of age, the course requires two doses rather than three, typically spaced one to six months apart depending on the clinical guidance followed.
Where to Get It
Private MenB vaccination is available through:
- Private GP practices and private clinic networks such as Bupa Health Clinics and Nuffield Health
- Travel health clinics, which routinely stock Bexsero and can administer it as part of a travel health consultation
- Some NHS GP practices which offer private vaccination services alongside their NHS work
- High street pharmacy chains including Boots and Lloyd's Pharmacy, which have offered private meningococcal vaccination at some locations
Availability varies by location and provider. It is worth calling ahead to confirm stock before booking. Your GP can provide a referral or recommendation if needed.
Cost
The cost of private MenB vaccination varies by provider. As a rough guide, expect to pay in the region of £100 to £150 per dose, making a full two-dose course approximately £200 to £300. Some providers charge a consultation fee in addition to the vaccine cost. Prices are not regulated and vary significantly, so it is worth comparing providers.
For families with more than one child outside the routine programme, the cumulative cost can be substantial. Some providers offer discounts for multiple family members vaccinated at the same appointment.
Who Should Particularly Consider It
Private MenB vaccination is worth discussing with a GP or healthcare provider for:
- Teenagers and young adults born before May 2015, particularly those approaching the 15 to 24 age peak
- Students starting university, where high carriage rates and close social mixing create the highest adult risk environment
- Anyone with a weakened immune system, for whom the consequences of invasive meningococcal disease are more severe
The JCVI's cost-effectiveness determination was a population-level policy judgement. At the individual level, the risk calculus is different. If the cost is manageable, vaccination is the only available protection against the most common cause of bacterial meningitis in the UK.
The UK Vaccination Programme in Full
For reference, the current NHS schedule as it relates to meningitis and related infections:
Infant Programme
MenB (Bexsero): introduced September 2015 for infants born on or after 1 May 2015. Currently given at 8 weeks, 12 weeks, and a booster at 12 months. The schedule has been periodically adjusted to provide earlier protection, most recently with the second dose moved to 12 weeks. Verify the current schedule at nhs.uk at time of publication.[2][3]
6-in-1 vaccine (DTaP/IPV/Hib/HepB): given at 8 weeks, 12 weeks, and 16 weeks. Includes Haemophilus influenzae type b (Hib), formerly a major cause of bacterial meningitis in young children.
Pneumococcal conjugate vaccine (PCV): given at 16 weeks and 12 months. Protects against Streptococcus pneumoniae, a significant cause of bacterial meningitis in adults and older people.
MMR: given at 12 months and 3 years 4 months. Mumps was the most common cause of viral meningitis before the MMR programme. Its near-elimination in vaccinated populations is one of the lesser-known successes of childhood immunisation.
Teenage Programme
MenACWY: offered in Year 9 (age 13 to 14). Also available as catch-up for anyone under 25 who missed it. Reduces carriage of the groups it covers and therefore provides herd protection. Does not cover MenB.
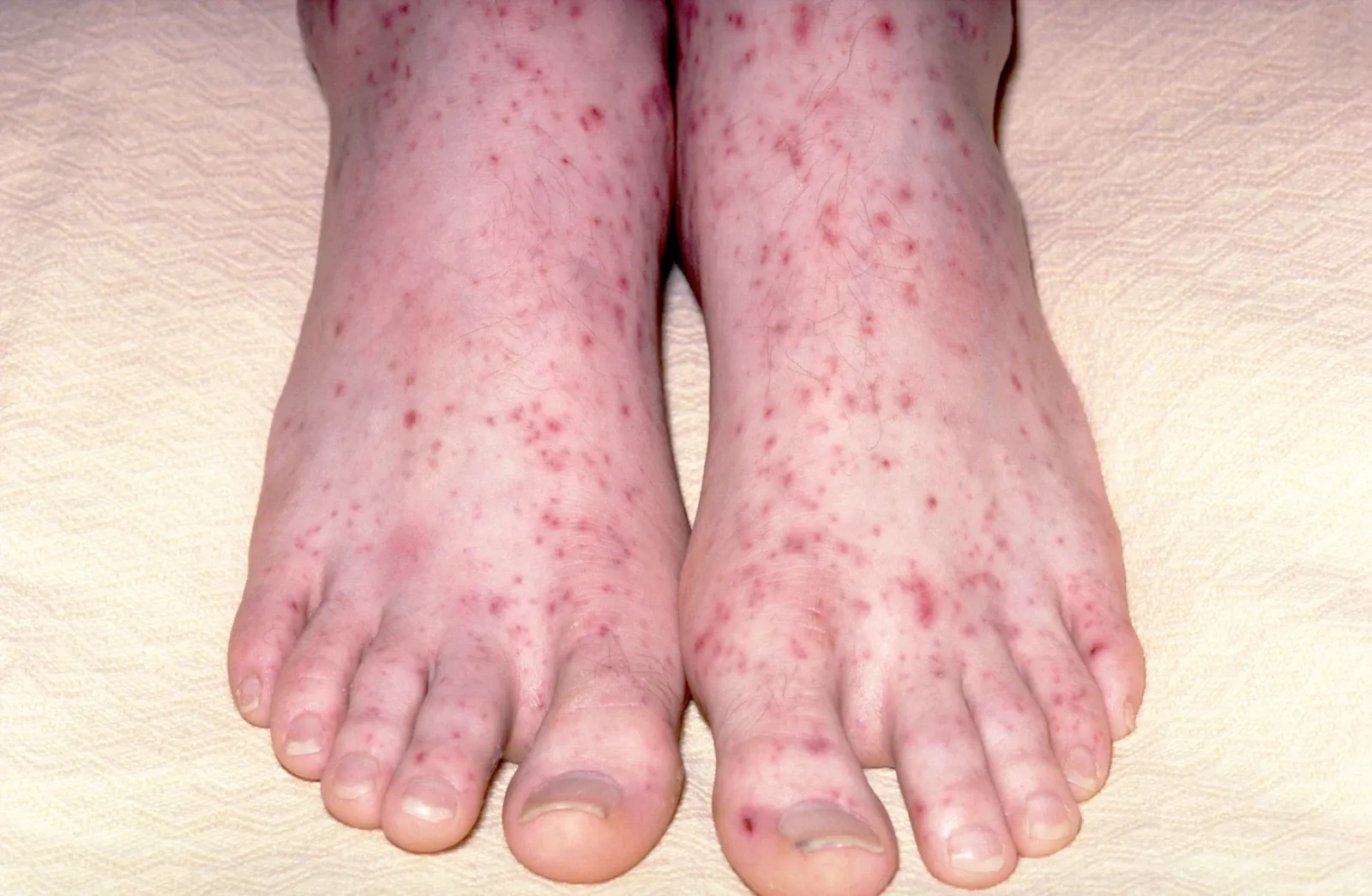
Recognising Meningitis: What You Need to Know
Understanding the full clinical picture is more useful than memorising a list of symptoms. The reason meningitis is dangerous is not that its signs are obscure. It is that they develop gradually, can be mistaken for other illnesses, and can progress to a life-threatening emergency with alarming speed.
- Confusion or difficulty waking
- Seizures with fever in someone with no history of epilepsy
- A non-fading rash
- Severe headache with vomiting that is rapidly worsening
- Any rapidly deteriorating illness, even without a rash
The Problem with Early Symptoms
In the early stages, meningitis often looks like flu. Fever, headache, muscle aches, and malaise are common early features. The classic triad of bacterial meningitis, consisting of sudden high fever, neck stiffness, and altered mental status, is present in only 44 to 46% of cases.[8] Waiting for all three to appear before seeking help is not a safe strategy.
What matters is trajectory. A person who is ill and getting worse, particularly one who is developing new symptoms or becoming increasingly unwell despite apparent treatment, needs urgent assessment. This is especially true in children, where the ability to report symptoms is limited and where the speed of deterioration can be faster than in adults.
Symptoms in Adults and Older Children
The following may appear in any combination and in any order:
- Sudden severe headache, often described as the worst the person has ever experienced
- High temperature, though occasionally meningitis presents with a low or normal temperature
- Stiff neck: the person is unable to touch their chin to their chest
- Sensitivity to light and loud sounds
- Nausea and vomiting, often projectile
- Confusion, drowsiness, or difficulty waking
- Seizures with fever in someone with no previous history
- Non-fading rash: red or purple spots that do not disappear under pressure
- Cold hands and feet, sometimes with a pale or blotchy appearance
Symptoms in Babies and Young Children
- High-pitched, unusual cry or moaning
- Refusing feeds or vomiting repeatedly
- Bulging fontanelle: the soft spot on top of the head may bulge outward
- Blank or staring expression, appearing unresponsive or difficult to rouse
- Dislike of being handled: screaming when picked up, a sign that movement causes pain
- Pale, blotchy, or mottled skin
- Fever, though some infants present without fever
- Non-fading rash
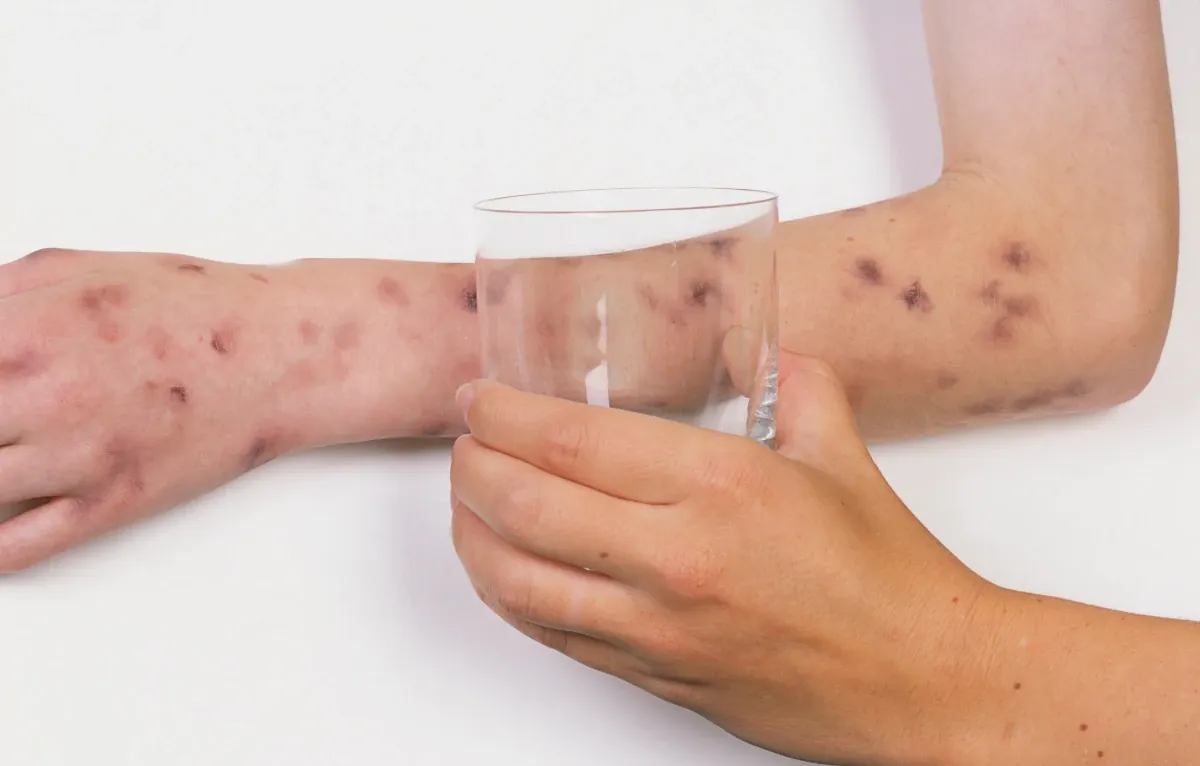
The Glass Test
The glass test involves pressing a clear glass firmly against a rash and observing whether the rash disappears under pressure. A rash that does not fade, visible through the glass, indicates blood has leaked into the skin: a sign of septicaemia and a medical emergency.
The rash typically appears late in the illness. By the time it is visible, the condition is already advanced.
A significant proportion of meningococcal cases never produce a rash at all.
The rash begins as tiny pin-prick spots (petechiae), which can be very difficult to see on both pale and darker skin tones. Check the entire body, including the inside of the eyelids and mouth.
Do not wait for a rash before calling 999. A rapidly deteriorating patient is an emergency regardless.
Long-Term Consequences of Meningitis
The severity of meningitis is measured not only in mortality but in what it leaves behind. Research from UCL's Institute of Child Health found that one in three children who survive meningococcal group B disease are left with after-effects.[10]
Hearing loss is the most common complication. It ranges from mild impairment to profound bilateral deafness. The mechanism is sensorineural: infection spreads into the cochlea, damaging the hair cells responsible for translating sound into electrical signals. Hearing loss may not be immediately apparent after recovery and can worsen in the weeks and months that follow. All survivors should have a formal hearing assessment.
Limb amputation and tissue damage result from purpura fulminans, the severe form of meningococcal septicaemia in which blood clots form in small vessels, cutting off circulation to the extremities. Fingers, toes, hands, and feet may require amputation. Extensive scarring from skin necrosis is also common.
Neurological effects include memory problems, learning difficulties, reduced concentration, and behavioural changes. These are often invisible and may not become apparent until a child returns to education. The MOSAIC study found that cognitive and psychological after-effects in school-age survivors were more common than previously recognised.[10]
Waterhouse-Friderichsen syndrome is a severe complication involving haemorrhagic destruction of the adrenal glands as a result of meningococcal septicaemia. It causes acute adrenal insufficiency, contributing to circulatory collapse, and is associated with high mortality.
Prevention: What Vaccination Can and Cannot Do
Vaccination is the single most effective tool against the most dangerous forms of meningitis, but it is worth being precise about what the current UK programme does and does not provide.
The MenB vaccine covers the substantial majority of MenB strains currently causing disease in England, but not all. The MenACWY vaccine provides good protection against four serogroups but not MenB. No current vaccine protects against viral causes of meningitis, other than indirectly through the MMR's effect on mumps. No vaccine protects against PAM. Vaccination reduces risk substantially but does not eliminate it.
Beyond vaccination, the following reduce transmission of bacterial meningitis:
- Not sharing food, drinks, cutlery, or cigarettes, which are the primary transmission routes through oral secretions
- Ensuring MenACWY vaccination before university, where close social mixing creates the highest adult risk environment
- Considering private MenB vaccination for teenagers and young adults outside the routine programme (see the dedicated section above)
- Maintaining awareness of symptoms so that early deterioration is recognised and acted on promptly
Meningitis kills and disables because of delay: delay in recognition, delay in seeking help, delay in treatment.
Why First Aid Training Matters
Every point in that chain where knowledge and confidence exist is a point where outcomes can be improved.
First aid training does not give you the ability to treat meningitis. It gives you the ability to recognise that something is seriously wrong, to act on that recognition without hesitation, and to provide appropriate support while emergency care is on its way. It normalises the decision to call 999, removes the paralysis that uncertainty creates, and instils an understanding of why deterioration in someone who is already unwell is itself an emergency sign.
In conditions like meningitis, where hours matter and the disease can move from early symptoms to irreversible damage within a single day, that trained recognition is not a small thing. It is often the difference between the story of a survival and the story of a death.
Recognising serious illness early is one of the most important skills first aid training provides.
If your workplace, school or organisation wants staff to feel confident responding to medical emergencies, you can learn more about our first aid training here.
References
1. Epidemiological Trends in Viral Meningitis in England: Prospective National Surveillance, 2013 to 2023. Journal of Infection (2024).
2. UKHSA. Invasive Meningococcal Disease in England: Annual Laboratory-Confirmed Reports for Epidemiological Year 2024 to 2025.
3. Giuliani MM et al. A universal vaccine for serogroup B meningococcus. Science (2006); and ScienceDirect: Introduction of Bexsero into the national infant immunisation programme (2015).
4. Ramsay ME et al. Herd immunity from meningococcal serogroup C conjugate vaccination in England. BMJ (2003).
5. Christensen H et al. Meningococcal carriage by age: a systematic review and meta-analysis. Lancet Infectious Diseases (2010).
6. Department of Health. MenB vaccination programme: eligibility and implementation guidance (September 2015).
7. Meningitis Now. No Plan B for MenB campaign documentation (meningitisnow.org). Accessed 2026.
8. van de Beek D et al. Clinical features and prognostic factors in adults with bacterial meningitis. New England Journal of Medicine (2004).
9. Brown TJ. In Hot Water. New Zealand Geographic (cited for NZ PAM case history and natural infection dynamics).
10. Viner RM et al. (UCL Institute of Child Health). MOSAIC Study: Outcomes of Serogroup B Meningococcal Disease in Children. The Lancet Neurology.
11. Capewell LG et al. Naegleria Infection and Primary Amebic Meningoencephalitis. StatPearls, NCBI Bookshelf (February 2025).
12. Health New Zealand | Te Whatu Ora. Meningoencephalitis: Primary Amoebic. Communicable Disease Control Manual (updated May 2012).





